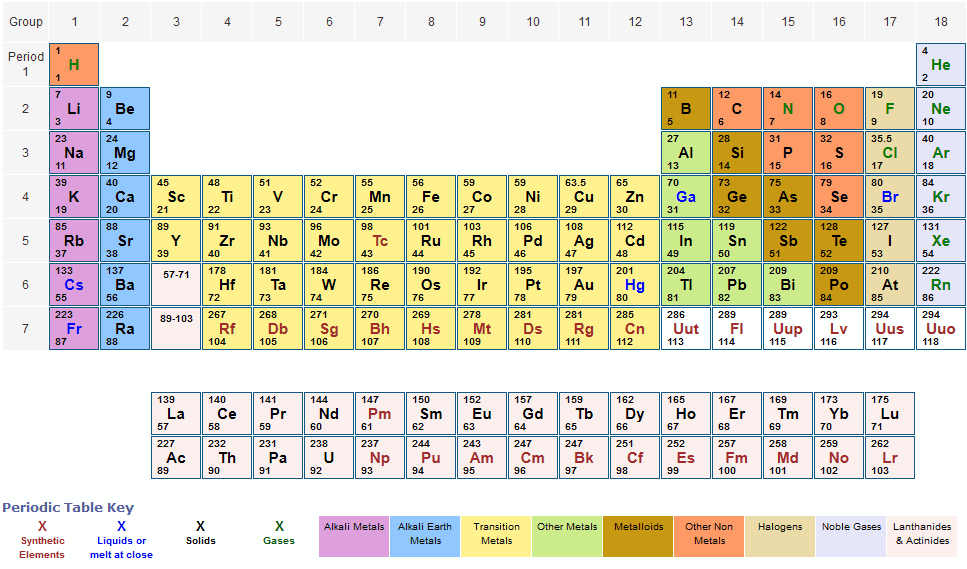
The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. We created a timeline of the history of the periodic table. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Who invented the Periodic Table of Elements? Molar mass of carbon: 12.01 g/mol Molar mass of hydrogen: 1.008 g/mol Step 4: For each element, multiply its molar mass by the. This table can be obtained in PDF format here. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. Orientation: what is physical chemistry about Part One. This periodic table contains the atomic masses of the elements as accepted by the IUPAC (09-2013). Buy print or eBook Opens in a new window Book contents. The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. Step 3: Find the molar mass of each element from the periodic table. Periodic table of the elements, with molar masses A Life Scientists Guide to Physical Chemistry. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? Every hydrogen atom has one proton in its nucleus. Compounds- carbon dioxide, sulphur dioxide, sulphur trioxide, nitric acid, ethane, acetic acid, sodium carbonate, sulphuric acid, calcium carbonate, hydrogen sulphide. Now, let me recall to you the atomic mass of lithium, sodium and potassium. The main thing I want to tell you is that he arranged these elements in the increasing order of their ATOMIC MASS. Hydrogen, at the upper left of the table, has an atomic number of 1. Answer: ELEMENTS-Boron, carbon, oxygen, iodine, argon, calcium, gold, silver, copper, zinc. Calcium (Ca), strontium (Sr) and Barium (Ba) in other triad and, Chlorine (Cl), bromine (Br) and iodine (I) in other triad. In this table, an elements atomic number is indicated above the elemental symbol. The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. The Periodic table of elements is a tool, developed by scientists over hundreds of years. 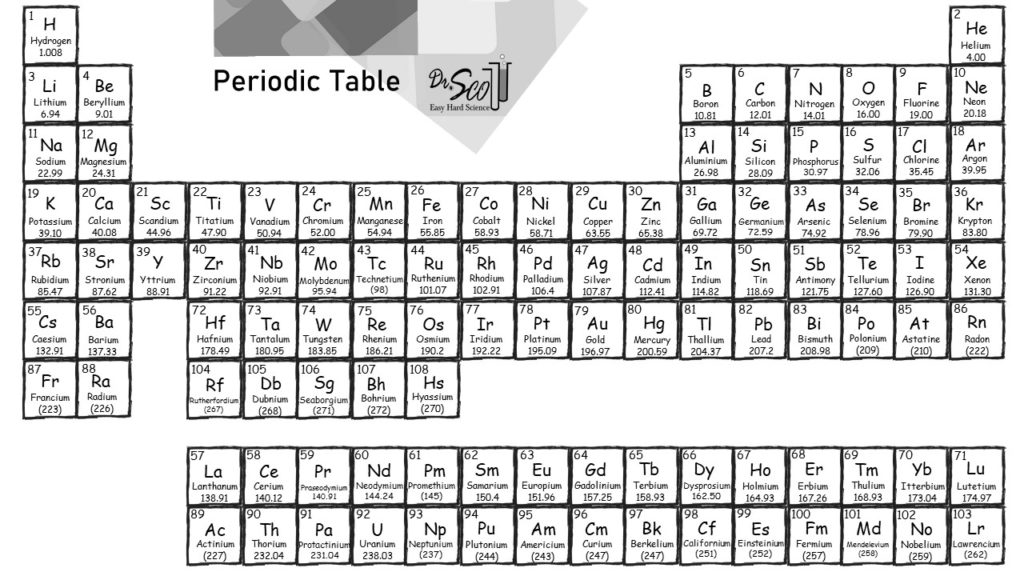 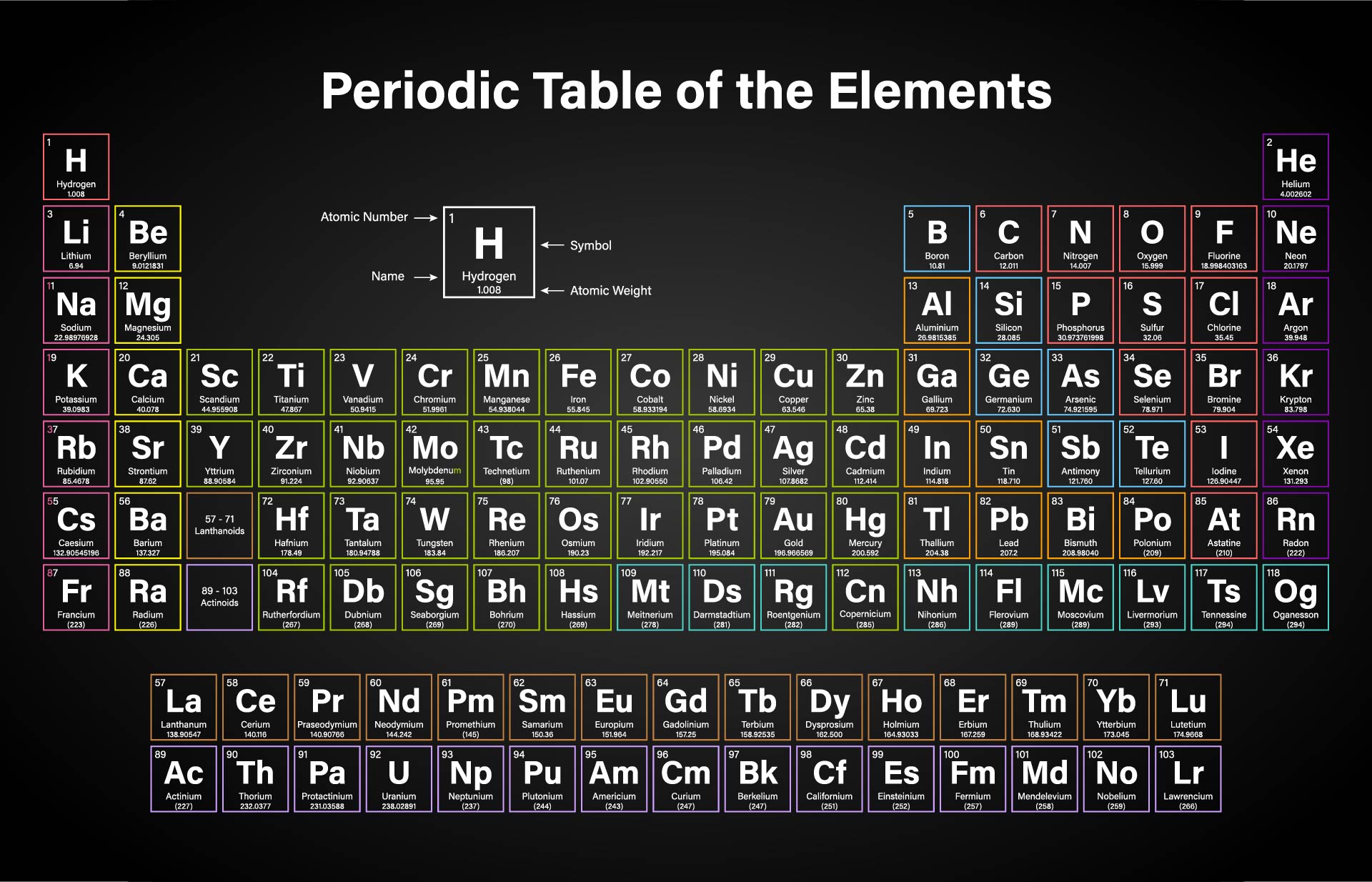
List of all the elements and their properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
